Abstract
Background: Elevated homocysteine levels are associated with increased blood coagulation and platelet activity and may modulate the response to antiplatelet therapies. We aimed to investigate the effects of homocysteine levels on the efficacy and safety of ticagrelor–acetylsalicylic acid (ASA) versus clopidogrel–ASA among patients with minor stroke or transient ischemic attack who carried CYP2C19 loss-of-function alleles.
Methods: We conducted a post hoc analysis of the CHANCE-2 (The Clopidogrel in High-risk Patients with Acute Nondisabling Cerebrovascular Events–II) trial. Participants were randomly assigned to treatment with ticagrelor–ASA or clopidogrel–ASA. We categorized participants into groups with elevated and non-elevated homocysteine levels, based on the median level. The primary efficacy outcome was recurrent stroke within 90-day follow-up. The primary safety outcome was severe or moderate bleeding within 90 days.
Results: A total of 2740 participants were randomly assigned to receive ticagrelor–ASA and 2700 to receive clopidogrel–ASA. Use of ticagrelor–ASA was associated with a reduced risk of recurrent stroke among participants with elevated homocysteine levels (74 [5.3%] v. 119 [8.5%]; hazard ratio [HR] 0.60, 95% confidence interval [CI] 0.45–0.81), but not among those with non-elevated levels (86 [6.4%] v. 87 [6.7%]; HR 0.97, 95% CI 0.71–1.32; p = 0.04 for interaction). When analyzed as a continuous variable, the benefits of ticagrelor–ASA with regard to recurrent stroke increased as homocysteine levels increased (p = 0.04 for interaction). No significant interaction between homocysteine levels and treatment with regard to severe or moderate bleeding was observed (p = 0.7 for interaction). We found a significant interaction between homocysteine levels and therapy with regard to recurrent stroke in females (p = 0.04 for interaction) but not males.
Interpretation: In comparison with clopidogrel–ASA, ticagrelor–ASA conferred more benefit to patients with elevated homocysteine levels, particularly to female patients, in this secondary analysis of a randomized controlled trial involving patients with minor ischemic stroke or TIA.
Trial registration: ClinicalTrials.gov, no. NCT04078737
Minor ischemic stroke or transient ischemic attack (TIA) are common disorders with a high rate of subsequent disabling stroke.1 Several trials have shown the benefit of dual antiplatelet therapy with clopidogrel–acetylsalicylic acid (ASA) or ticagrelor–ASA in the secondary prevention of stroke.2–4 However, despite antiplatelet therapy, the risk of another stroke within 3 months after the initial event is still approximately 5%–10%,5–7 which might be owing to the variability of patient responses to different antiplatelet therapies, with a considerable proportion exhibiting diminished or absent antiplatelet effects.8,9 Early identification and clinical management of these patients is thus important.
Levels of homocysteine, a metabolite of methionine degradation, are associated with the progression of atherosclerosis.10,11 Elevated levels of homocysteine induce changes in coagulation, tissue factor expression, fibrinolysis, and endothelial function, and also impair the function of platelets, thereby increasing the risk of initial and recurrent stroke.12–14 Additionally, elevated levels of homocysteine could enhance the metabolism of eicosanoids and promote the overproduction of platelet thromboxane, leading to increased platelet adhesion and inadequate platelet inhibition.15
The hypothesis that homocysteine levels could modulate platelet reactivity in patients treated with dual antiplatelet medications has been tested in several trials conducted among patients with acute coronary syndrome undergoing percutaneous coronary interventions, with contrasting results.16–19 Little is known about whether homocysteine levels could modify the benefits of different dual antiplatelet treatments in preventing stroke.
We aimed to investigate the effect of homocysteine levels on the efficacy and safety of ticagrelor–ASA versus clopidogrel–ASA among patients with minor ischemic stroke or TIA who carried CYP2C19 loss-of-function (LOF) alleles, using data from the CHANCE-2 (Clopidogrel in High-risk Patients with Acute Nondisabling Cerebrovascular Events–II) trial.
Methods
We conducted a post hoc analysis of the CHANCE-2 trial. Details on the study design, protocol, and primary results of the CHANCE-2 trial have been published elsewhere.4,20 Briefly, the CHANCE-2 trial was a multicentre, randomized, double-blind, placebo-controlled trial designed to assess the efficacy and safety of dual-antiplatelet therapy with ticagrelor–ASA versus clopidogrel–ASA in CYP2C19 LOF carriers with stroke or TIA. Inclusion criteria were patients who were aged 40 years or older, had either an acute nondisabling stroke with a National Institutes of Health Stroke Scale score of 3 or less or a high-risk TIA with an ABCD2 (Age, Blood pressure, Clinical features, Duration of symptoms, Diabetes) score of 4 or greater, and could start the trial within 24 hours after symptom onset. A total of 6412 patients were enrolled at 202 centres in China from Sept. 23, 2019, to Mar. 22, 2021. For this analysis, we excluded 972 patients with missing data on homocysteine.
Randomization and treatment
Within 24 hours after symptom onset, patients were randomly assigned in a 1:1 ratio to receive 90 days of ticagrelor (180 mg loading dose on day 1 followed by 90 mg twice daily on days 2–90 plus placebo clopidogrel) or clopidogrel (300 mg loading dose on day 1 followed by 75 mg daily on days 2–90 plus placebo ticagrelor). All patients received 21 days of ASA (75 to 300 mg loading dose on day 1 followed by 75 mg daily for 21 days).
Measurements of homocysteine
Fasting venous blood samples were drawn in serum separator tubes within 48 hours of symptom onset for the biochemical measurements. Serum was extracted within 1 hour and shipped on ice by overnight courier from each participating hospital to Beijing Tiantan Hospital (China), where all data analyses were performed. The specimens were stored in a −80°C frost-free freezer and were not thawed until analyzed for this study. Levels of total homocysteine were measured with the enzymatic method in the clinical laboratory in Beijing Tiantan Hospital by laboratory personnel unaware of the clinical data. The intra-assay coefficient of variation of homocysteine concentrations was 2.7%.
Efficacy and safety outcomes
The primary efficacy outcome was recurrent stroke within 90 days. The secondary efficacy outcomes included new stroke within 30 days, a vascular event (a composite of stroke, TIA, myocardial infarction or vascular death) within 90 days, ischemic stroke within 90 days, disabling stroke (Modified Rankin Scale [MRS] score ≥ 2) at 90 days, and severity of stroke or TIA21 within 90 days (a 6-level ordinal scale to measure severity: fatal stroke, severe stroke [stroke with subsequent MRS score of 4 or 5], moderate stroke [stroke with subsequent MRS score of 2 or 3], mild stroke [stroke with subsequent MRS score of 0 or 1], TIA, and no stroke or TIA).
The primary safety outcome was severe or moderate bleeding, which was defined using the Global Utilization of Streptokinase and Tissue Plasminogen Activator for Occluded Coronary Arteries criteria within 90 days.22 Secondary safety outcomes included any bleeding, death, adverse events, and severe adverse events through 90 days of follow-up. All efficacy and safety outcomes were confirmed by an independent clinical event adjudication committee, whose members were unaware of which treatment patients received.
Statistical analysis
We analyzed homocysteine levels as both categorical and continuous variables. The threshold of homocysteine levels that predict atherosclerotic diseases has varied in different regions and populations, and no specific cutoff has been found in previous studies.23–26 Therefore, we used the median homocysteine value (12.9 μmol/L) to define groups with elevated and non-elevated homocysteine levels, as previously done.27–30 We presented descriptive data as medians with interquartile ranges (IQR) for continuous variables and frequencies with percentages for categorical variables. We compared baseline characteristics between ticagrelor–ASA and clopidogrel–ASA in different homocysteine categories by Wilcoxon test for continuous variables and by χ2 test for categorical variables. We used the Kaplan–Meier product limit method to generate survival plots on the primary efficacy outcome.
We assessed differences in the primary outcome and other survival outcomes during the 90-day follow-up period using a Cox proportional hazards regression model with study centres set as a random effect; we reported hazard ratios (HRs) with 95% confidence intervals (CIs). When there were multiple events of the same type, we used the time to the first event in the model. We censored data from patients who had no event during 90-day follow-up at termination of the trial or nonvascular death. We tested the proportional hazards assumption using Schoenfeld residuals, which was met for all models (p > 0.05 for all models, Appendix 1, Table S1, available at www.cmaj.ca/lookup/doi/10.1503/cmaj.231262/tab-related-content). We performed shift analysis for comparison of the secondary outcome of ordinal stroke or TIA between the 2 treatment groups with the use of logistic regression, and we calculated the common odds ratio and 95% CI. We evaluated interactions of treatment assignment with homocysteine categories with the addition of treatment by homocysteine categories in the model. We also evaluated the impact of homocysteine levels as a continuous variable on the effect of dual-antiplatelet therapy on recurrent stroke, assuming a linear relation. Because renal disease may change homocysteine levels, we performed a sensitivity analysis adjusted for renal function. Finally, we performed subgroup analyses stratified by sex and CYP2C19 genotypes.31,32
We performed all statistical analyses with SAS statistical software, version 9.4 (SAS Institute Inc.). All tests were 2-sided, and we considered a p value less than 0.05 to be significant.
Ethics approval
The trial was approved by the ethics committee at Beijing Tiantan Hospital (approval no. KY2019-035-02) and all participating centres. Written informed consent was provided by all the patients or their representatives before enrolment.
Results
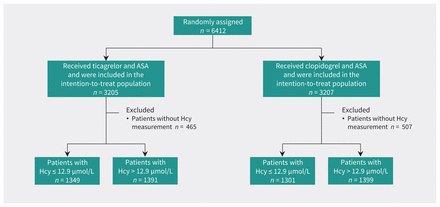
A total of 5440 participants were entered into the study. The number of patients randomly assigned to the 2 treatment groups, patients excluded because of missing homocysteine levels, and patients with low and high homocysteine levels are shown in Figure 1. Compared with patients excluded, those who were included were more likely to be younger, male, current smokers, randomly assigned more than 12 hours after symptom onset, and to have a qualifying TIA and to have had previously taken antiplatelet therapy. There was no difference in the rate of recurrent stroke within 90 days between the included and excluded patients (Appendix 1, Table S2).
Flow chart of the study. Note: ASA = acetylsalicylic acid, Hcy = homocysteine.
The median homocysteine level of patients included in this analysis was 12.9 μmol/L (IQR 10–17.98 μmol/L); it was higher among males (14.1 μmol/L, IQR 10.9–20.0 μmol/L) than females (11.1 μmol/L, IQR 8.8–14.6 μmol/L). Baseline characteristics were not significantly different between the ticagrelor–ASA and clopidogrel–ASA groups within homocysteine categories, except that patients with elevated homocysteine were older in the ticagrelor–ASA group than those in the clopidogrel–ASA group (median difference 0.7 yr) (Table 1).
Baseline characteristics according to homocysteine level and treatment
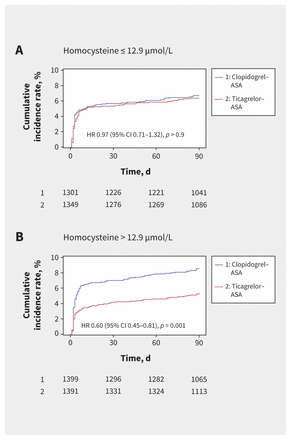
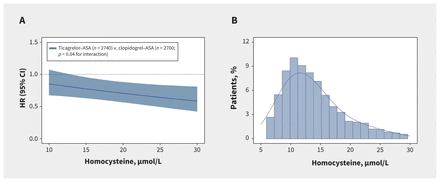
Use of ticagrelor–ASA significantly reduced the risk of recurrent stroke within 90 days among patients with elevated homocysteine levels (74 [5.3%] v. 119 [8.5%]; HR 0.60, 95% CI 0.45–0.81; p < 0.001), whereas this benefit was not seen in patients with non-elevated homocysteine levels (86 [6.4%] v. 87 [6.7%]; HR 0.97, 95% CI 0.71–1.32; p = 0.8), with a significant homocysteine category-by-treatment interaction of 0.04 (Table 2 and Figure 2). When analyzed as a continuous variable, homocysteine levels also significantly modulated the effect of ticagrelor–ASA on the primary outcome. As homocysteine levels increased, the risk of recurrent stroke within 90 days decreased among patients receiving ticagrelor–ASA compared with those receiving clopidogrel–ASA (p = 0.04 for interaction) (Figure 3). Similar trends were seen with the secondary outcomes, although the test for interaction was not significant (Table 2). Sensitivity analysis adjusted for renal function yielded similar results (Appendix 1, Table S3).
Efficacy and safety outcomes of patients according to homocysteine level and treatment
Cumulative probability of stroke according to homocysteine levels and treatment. Note: ASA = acetylsalicylic acid, CI = confidence interval, HR = hazard ratio.
Effects of ticagrelor–ASA versus clopidogrel–ASA on the primary efficacy outcome by homocysteine level as a continuous variable and distribution of homocysteine levels. (A) The linear association between homocysteine level and hazard ratio for the primary efficacy outcome. The significant interaction test indicates that the slope is significantly different from zero. (B) The distribution of homocysteine levels. Note: ASA = acetylsalicylic acid, CI = confidence interval, HR = hazard ratio.
The primary safety outcome of severe or moderate bleeding occurred with similar frequency in the ticagrelor–ASA group and clopidogrel–ASA group independent of homocysteine levels (0.2% v. 0.2% in the group with elevated homocysteine levels; 0.4% v. 0.4% in the group with non-elevated homocysteine levels; p = 0.7 for interaction). Homocysteine category did not affect the risk of other safety outcomes (Table 2).
Among female patients, treatment with ticagrelor–ASA, compared with clopidogrel–ASA, was associated with a lower rate of recurrent stroke only in those with elevated homocysteine levels (HR 0.45, 95% CI 0.22–0.90; p = 0.007 for interaction). Use of ticagrelor–ASA reduced recurrent stroke to a similar degree in male patients regardless of homocysteine levels (p = 0.8 for interaction; Appendix 1, Table S4). Sex differences were also observed for secondary outcomes (Appendix 1, Table S4). There was no significant interaction between homocysteine levels and antiplatelet therapy among patients who were intermediate and low metabolizers (Appendix 1, Table S5).
Interpretation
We found that, compared with clopidogrel–ASA, ticagrelor–ASA significantly reduced the risk of recurrent stroke without increased bleeding events among patients who carried the CYP2C19 LOF alleles who had minor stroke or TIA and elevated homocysteine levels, especially female patients. In this post hoc analysis, we found no difference between the 2 treatments among patients with non-elevated homocysteine levels. The results suggest that homocysteine could be a potential biomarker to guide dual-antiplatelet strategies, but these results should be replicated by others before they influence practice.
Studies have shown that homocysteine is a strong enhancer of platelet reactivity in vivo and in vitro,33,34 possibly by promoting the activation of factor V, suppressing thrombomodulin and protein C, interfering with the normal balance of coagulation, blunting endothelial function, enhancing oxidative stress, inducing inflammation, enhancing adhesion of thrombin-activated platelets to collagen and fibrinogen, and promoting the production of platelet thromboxanes. 15,18,35 However, studies of the effects of homocysteine levels on platelet reactivity in patients receiving antiplatelet therapies have found inconsistent results. One randomized multicentre study showed that among patients with coronary artery disease undergoing coronary angiography, elevated homocysteine was an independent predictor of suboptimal response to ASA.19 Similar results were reported by the Novara Atherosclerosis Study Group.18 However, another study reported that homocysteine levels did not significantly affect the rate of on-treatment platelet reactivity in patients treated with ticagrelor–ASA, or with adenosine–diphosphate antagonists (mainly clopidogrel).17 These studies did not address the possible role of homocysteine in modifying the response to antiplatelet therapy by testing the interaction between homocysteine levels and antiplatelet treatments, and none of them studied patients with stroke.
Although the primary results of the Vitamins to Prevent Stroke (VITATOPS) trial36 and Vitamin Intervention for Stroke Prevention (VISP) trial37 did not provide evidence that lowering homocysteine levels with folate and B vitamin therapy (including folic acid, vitamin B6, and vitamin B12) was effective for secondary prevention of stroke, post hoc analyses showed the effects of homocysteine-lowering agents on outcomes differed by antiplatelet usage. The post hoc analysis of the VITATOPS trial showed a significant interaction between B vitamins and antiplatelet therapy.38 Of the participants taking antiplatelet drugs at baseline, B vitamins had no significant effect on the risk of recurrent stroke, but a significant effect was seen in participants not taking antiplatelet drugs. Similarly, the post hoc analysis of the VISP trial showed that high-dose vitamin therapy was associated with higher stroke risk among patients taking antiplatelet drugs, but trended toward lower risk among those not taking antiplatelet drugs.39 These findings were further confirmed by a meta-analysis including 11 randomized controlled trials.40 Because the benefits of homocysteine lowering on the risk of recurrent stroke were modified by antiplatelet therapies, it is possible that the benefits of antiplatelet therapy may differ by homocysteine levels. This hypothesis was supported by the post hoc analysis of the CHANCE trial, showing that among female patients with minor stroke or high-risk TIA, non-elevated homocysteine levels were associated with better response to dual antiplatelet therapy with clopidogrel–ASA than monotherapy with ASA.27 Our study adds to this information by showing that elevated homocysteine levels were associated with better response to ticagrelor–ASA therapy than clopidogrel–ASA therapy in CYP2C19 LOF carriers with minor stroke or TIA.
A possible mechanism underlying our finding may be a synergistic relation between the thrombotic effects of elevated homocysteine levels and the antithrombotic effects of dual antiplatelet treatments. Laboratory investigations suggested that elevated homocysteine could impair vascular function in the absence of adequate and effective antiplatelet therapies through potential mechanisms including impairment of endothelial function, oxidation of low-density lipids, increased monocyte adhesion to the blood vessel wall, increased lipid uptake and retention, activation of inflammatory pathways, stimulatory effects on smooth-muscle-cell proliferation, and prothrombotic tendency mediated by activation of coagulation factors and platelet dysfunction.41–43 The prothrombotic condition in CYP2C19 LOF carriers with elevated homocysteine levels may require a high degree of platelet inhibition to provide adequate protection against ischemic events. Ticagrelor, which does not require metabolic activation, may be superior to clopidogrel by directly inhibiting platelet activation and thrombus formation and indirectly reducing vasoconstrictor tone, vascular smooth-muscle-cell proliferation, and release of inflammatory cytokines, oxygen radicals, growth factors leading to platelet activation and thrombus formation among CYP2C19 LOF carriers.4,44
Homocysteine levels were lower in females than in males, and our analyses found that female patients had a better response to ticagrelor–ASA therapy if homocysteine levels were elevated. This finding is in accordance with the post hoc analysis of the CHANCE trial, in which female patients did not respond to clopidogrel–ASA therapy if homocysteine levels were elevated.27 Females have higher platelet reactivity, higher prevalence of stroke, and poorer prognosis after stroke than males.45,46
Limitations
We did not have information on diet, and folate concentration may have influenced the levels of homocysteine before the index event and during follow-up. However, since the trial was conducted in China, a country with no mandatory fortification of foods with folic acid or widespread use of folic acid supplements, the magnitude of fluctuation of homocysteine levels was likely to be low. The incidence of bleeding events was low in the trial, which may reduce our statistical power to detect an interaction between homocysteine levels, antiplatelet treatments, and bleeding. We excluded about 15% of patients because of missing data on homocysteine levels; however, there were no large differences in baseline characteristics between those excluded and included in the study. Finally, this was a post hoc analysis; our findings should be considered hypothesis generating and need to be confirmed by other studies.
Conclusion
We found that among patients who carried the CYP2C19 LOF alleles who had minor stroke or TIA, use of ticagrelor–ASA, compared with clopidogrel–ASA, was associated with a reduced risk for recurrent stroke without an increase in severe or moderate bleeding events among patients with elevated homocysteine levels. Patients with non-elevated homocysteine levels did not derive the same benefit from ticagrelor–ASA. These results should be confirmed in other studies.
Acknowledgments
The authors thank all study participants, their relatives, and the members of the survey teams at the 202 centres of the CHANCE-2 study.
Footnotes
Competing interests: Philip Bath reports study grants from the British Heart Foundation and National Institute for Health and Care Research Health Technology Assessment; consulting fees from CoMind, DiaMedica, Phagenesis, and Roche; support for attending meetings or travel from Brainomix; and participation on a board for European Carotid Surgery Trial 2 and AVRT (atrioventricular reciprocating tachycardia) Dose. Dr. Bath reports being co-chair of the Industry Committee of the World Stroke Organisation and receipt of trial devices from Phagenesis for the Pharyngeal Electrical stimulation for Acute Stroke dysphagia Trial. No other competing interests declared.
This article has been peer reviewed.
Contributors: Anxin Wang, Xue Tian, Xia Meng, and Yongjun Wang contributed to the conception and design of this work. Anxin Wang, Xue Tian, Xia Meng, Xuewei Xie, Hao Li, Philip M. Bath, Jing Jing, Jinxi Lin, Yilong Wang, Xingquan Zhao, Zixiao Li, and Liping Liu contributed to the acquisition, analysis, and interpretation of data. Anxin Wang, Xue Tian, and Xia Meng drafted the manuscript. All of the authors revised the manuscript critically for important intellectual content, gave final approval of the version to be published, and agreed to be accountable for all aspects of the work. Anxin Wang and Xue Tian contributed equally to this work. Anxin Wang and Xia Meng were the co–corresponding authors.
Funding: This work was supported by the National Key Research and Development Program of China (2022YFC3600600 and 2022YFC3600400), Beijing Natural Science Foundation Haidian Original Innovation Joint Fund (L222123), Fund for Young Talents of Beijing Medical Management Center (QML20230505), the high-level public health talents (xuekegugan-02-47), and Training Fund for Open Projects at Clinical Institutes and Departments of Capital Medical University (CCMU2022ZKYXZ009). Salubris contributed ticagrelor, clopidogrel, and placebo at no cost and with no restrictions. Chongqing Jingyin Bioscience Co., Ltd. provided GMEX point-of-care genotyping system and technical supports for CHANCE-2 at no cost and with no restrictions.
Data sharing: Data are available to researchers on request for purposes of reproducing the results or replicating the procedure by directly contacting the corresponding authors.
- Accepted December 4, 2023.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use), and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/