Abstract
Background: The COVID-19 pandemic has had a major impact on access to health care resources. Our objective was to estimate the impact of the COVID-19 pandemic on the incidence of childhood cancer in Canada. We also aimed to compare the proportion of patients who enrolled in clinical trials at diagnosis, presented with metastatic disease or had an early death during the first 9 months of the COVID-19 pandemic compared with previous years.
Methods: We conducted an observational study that included children younger than 15 years with a new diagnosis of cancer between March 2016 and November 2020 at 1 of 17 Canadian pediatric oncology centres. Our primary outcome was the monthly age-standardized incidence rates (ASIRs) of cancers. We evaluated level and trend changes using interventional autoregressive integrated moving average models. Secondary outcomes were the proportion of patients who were enrolled in a clinical trial, who had metastatic or advanced disease and who died within 30 days. We compared the baseline and pandemic periods using rate ratios (RRs) and 95% confidence intervals (CIs).
Results: Age-standardized incidence rates during COVID-19 quarters were 157.7, 164.6, and 148.0 per million, respectively, whereas quarterly baseline ASIRs ranged between 150.3 and 175.1 per million (incidence RR 0.93 [95% CI 0.78 to 1.12] to incidence RR 1.04 [95% CI 0.87 to 1.24]). We found no statistically significant level or slope changes between the projected and observed ASIRs for all new cancers (parameter estimate [β], level 4.98, 95% CI −15.1 to 25.04, p = 0.25), or when stratified by cancer type or by geographic area. Clinical trial enrolment rate was stable or increased during the pandemic compared with baseline (RR 1.22 [95% CI 0.70 to 2.13] to RR 1.71 [95% CI 1.01 to 2.89]). There was no difference in the proportion of patients with metastatic disease (RR 0.84 [95% CI 0.55 to 1.29] to RR 1.22 [0.84 to 1.79]), or who died within 30 days (RR 0.16 [95% CI 0.01 to 3.04] to RR 1.73 [95% CI 0.38 to 15.2]).
Interpretation: We did not observe a statistically significant change in the incidence of childhood cancer, or in the proportion of children enrolling in a clinical trial, presenting with metastatic disease or who died early during the first 9 months of the COVID-19 pandemic, which suggests that access to health care in pediatric oncology was not reduced substantially in Canada.
Concerns have been raised that the COVID-19 pandemic disrupted health care–seeking behaviours and access to health care, affecting the diagnosis and management of other conditions such as cancer. Studies conducted in the Netherlands and United Kingdom using administrative data have shown as much as a 50% reduction in cancer incidence in adults after March 2020.1,2 Other studies in adult populations thus far have shown a decrease in the number of new cancer diagnoses, and cancer-related medical visits, therapies and surgeries, 1,3–5 raising concerns about potential excess cancer mortality in the upcoming years.6 This may be explained partly by the suspension or reduction of cancer-screening procedures, such as mammography, colonoscopy and cervical cytology by up to 90%,3,5,7 because these screening initiatives play a critical role in the detection of cancers in adults. A 2020 retrospective single-centre cohort study in Japan that involved 123 patients with colorectal cancer reported that significantly more of these patients presented with complete intestinal obstruction, which suggests that detection delays might have contributed to diagnosis at later stages of the disease.8 It is unclear whether these findings apply to childhood cancer because cancer screening is not part of routine pediatric care, and early detection may not be as important in childhood cancer than in its adult counterpart.9
In children, case series and single-centre retrospective cohort studies, notably from Italy and the United States, suggested a marked reduction in incident cancers, along with high acuity of care at presentation.10–13 Similar concerns of delayed clinical presentation were raised in other pediatric patient populations, with reports of children presenting at late stages of sepsis or diabetic ketoacidosis, which suggests a delay in seeking care.14,15
It is possible that fear of COVID-19 dissuaded families with children from seeking care for nonspecific symptoms such as pain, headache or fatigue, which are typical triggers leading to a pediatric cancer diagnosis. Understanding the indirect effects of health policies during the COVID-19 pandemic is important to guide policy-making and mitigate barriers to essential health care in future public health crises.
Our objective was to measure the impact of the COVID-19 pandemic and associated restrictions on the incidence of childhood cancer in Canada. We also aimed to compare the proportion of patients who enrolled in clinical trials at diagnosis, presented with metastatic disease or died during the first 9 months of the COVID-19 pandemic compared with previous years.
Methods
We conducted a retrospective interrupted time series analysis using a population-based active cancer registry (Cancer in Young People in Canada [CYP-C]).
Study setting
In Canada, pediatric oncology care is regionalized in 17 pediatric hematology–oncology centres, distributed across the country, which form the C17 Council (Appendix 1, available at www.cmaj.ca/lookup/doi/10.1503/cmaj.210659/tab-related-content).
Data sources
The CYP-C is a collaboration between the Public Health Agency of Canada, the C17 Council and the Canadian Partnership Against Cancer. Trained clinical research assistants or data managers abstract data prospectively from medical records at each site. Multiple approaches are used to ensure high-quality data.16–18 Data from the 5 Ontario sites are collected into the Pediatric Oncology Group of Ontario Networked Information System (POGONIS) and are transferred to CYP-C.
For our study, all centres contributed to accelerated data collection to obtain contemporary data and entered all new cancer diagnoses up to Nov. 30, 2020, into CYP-C as of June 22, 2021. The CYP-C Management Committee and the Pediatric Oncology Group of Ontario granted approval to use data in CYP-C for the purpose of this study.
Study population
We included all patients entered into CYP-C who were younger than 15 years of age at cancer diagnosis (aligned with CYP-C eligibility criteria), diagnosed with a neoplasm included in the International Classification of Childhood Cancer (ICCC-3), third edition, 19 and diagnosed and received treatment at 1 of the 17 pediatric oncology centres during the study periods. More than 95% of patients aged younger than 15 years at diagnosis in Canada receive treatment in 1 of these hospitals.17,20 We defined the COVID-19 pandemic period as March to November 2020 to correspond with the first reports of local transmission within Canada.21 The pre-pandemic, baseline period was December 2017 to February 2020.
Outcomes
Our primary outcome was the age-standardized incidence rate (ASIR) of newly diagnosed cancers per million population. We used the date of the most definitive diagnostic procedure, typically biopsy, as the date of diagnosis. Secondary outcomes included the proportion of patients who were enrolled in a clinical trial at diagnosis, presented with metastatic disease or died within 30 days of cancer diagnosis (early death). We considered enrolment in a clinical trial an important outcome because improvements in childhood cancer outcomes are thought to be largely attributable to high participation rates in cooperative clinical trials,22 whereas metastatic disease and early deaths were considered as proxies of delayed presentation or management. For solid cancers, any distant extension was considered metastatic disease. We categorized acute lymphoblastic leukemia as no central nervous system (CNS) involvement (CNS 1) or CNS positive (CNS 2 or 3). Precursor B-cell acute lymphoblastic leukemia was categorized as standard or high risk using the National Cancer Institute (NCI) risk scale.23 The presence of metastatic disease at presentation, CNS status and enrolment in a clinical trial are explicitly collected by CYP-C and POGONIS. We only considered enrolment in a therapeutic clinical trial at the initial diagnosis (excluding trials for supportive care interventions and for relapsed or refractory disease). Early death included all causes of death within 30 days of cancer diagnosis.
Statistical analysis
To compare quarterly outcomes, we compared pediatric cancers diagnosed from December 2017 to February 2020 (baseline period) to pediatric cancers diagnosed between March and November 2020 (COVID-19 pandemic). Then, to evaluate the association between the pandemic and changes in the monthly ASIR, we used cancer diagnoses between March 2016 and November 2020.
We calculated ASIRs using the direct method to standardize the age-specific rates to the 2011 Canadian population. Monthly and quarterly population estimates were based on the quarterly population estimates by age and sex between 2016 and 2020, for all Canadian provinces and territories.24,25
We defined each pandemic quarter incidence rate ratio (RR) as the quarterly ASIR divided by the median of ASIRs across the quarters of the baseline period. We estimated the 95% confidence interval (CI) of the incidence RR using the method for the ASIR.26
To evaluate changes in the monthly observed ASIRs during the pandemic period compared with the projected ASIRs, we performed interrupted time series analysis using the autoregressive integrated moving average model.27 The COVID-19 impact was evaluated by the changes in the level (a step change) and trend (slope) of the ASIRs.27–30 The methodology is detailed in Appendix 1.27,31
We performed stratified analysis by sex, cancer type and geographic area. We categorized cancer types as leukemia and lymphoma, CNS tumours or extracranial solid tumours19 because the diagnosis of some cancer types might require additional diagnostic and imaging procedures and thus may be more affected by the pandemic. Extracranial solid tumours included all non-CNS solid tumours, such as hepatic, renal and bone tumours.19
We divided Canada into 5 geographic regions (British Columbia, Prairies [Manitoba, Saskatchewan and Alberta], Ontario, Quebec and Atlantic provinces [Nova Scotia, Prince Edward Island, New Brunswick, Newfoundland and Labrador]) to reflect their differential burden of COVID-19. As of Aug. 1, 2020, the number of cumulative COVID-19 cases per 100 000 population for British Columbia, the Prairies, Ontario, Quebec and Atlantic provinces was 71.8, 182.0, 282.5, 700.8 and 63.5, respectively.32
We used RRs and 95% CIs to compare differences in proportions for all secondary outcomes between pandemic and baseline periods. We defined the RR as the ratio of the proportion for the pandemic quarter to the median of proportions in the baseline period. Whereas the asymptotic confidence limits for the RR were estimated for a large sample, we used exact unconditional confidence limits for RR based on the relative risk score statistic when the count was less than 5. When the count was zero, we computed an adjusted RR, a logit estimate of the RR and CI, using a 0.5 correction factor in each cell of 0.
All analyses were conducted using SAS (version 9.4). We considered p values of less than 0.05 to be significant, without adjustment for multiplicity. We used complete case analysis.
Ethics approval
The Pediatric Oncology Group of Ontario is a prescribed entity pursuant to Section 45 of the Ontario Personal Health Information Protection Act and, therefore, research ethics board approval was not required for the 5 Ontario sites. Collection of data in CYP-C was approved by the Research Ethics Boards of all participating sites outside Ontario with a waiver of the requirement for informed consent. The CYP-C Management Committee and the Pediatric Oncology Group of Ontario granted approval to use data in CYP-C for the purpose of this study. As we used aggregate data, approval was deemed unnecessary after evaluation by the CHU de Québec’s Research Ethics Board.
Results
During the COVID-19 pandemic quarters (March–May, June–August and September–November 2020), the ASIRs of new cancer diagnoses registered in CYP-C were 157.7, 164.6 and 148.0 per million population (Table 1). These estimates were comparable to ASIRs from December 2017 to February 2020 (median 158.4, interquartile range [IQR] 155.6 to 170.6), with incidence RRs of 1.00 (95% CI 0.83 to 1.19), 1.04 (95% CI 0.87 to 1.24) and 0.93 (95% CI 0.78 to 1.12) for each of the COVID-19 pandemic quarters, respectively.
Quarterly age-standardized incidence rates per million among Canadian children younger than 15 years of age who had a new diagnosis of cancer between December 2017 and November 2020
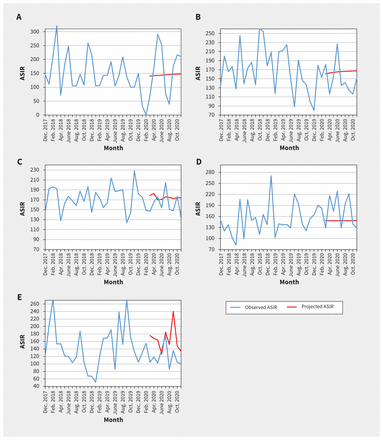
We found that the quarterly ASIRs of new cancer diagnoses were comparable to baseline values for both males and females (Table 1 and Appendix 1, Supplementary Table S1). We did not find statistically significant change in level and trend between projected and observed monthly ASIRs of new cancer diagnoses overall, when stratified by sex, cancer type (Table 2, Figure 1) or geographical area (Table 2 and Figure 2).
Results for the interventional autoregressive integrated moving average model summarizing the association of the COVID-19 pandemic and monthly age-standardized incidence rates for new pediatric cancer diagnoses per million population
Observed and projected monthly age-standardized incidence rates (ASIRs) of new pediatric cancer diagnoses per million, overall and stratified by cancer type: A) all cancers combined, B) leukemia and lymphoma, C) central nervous system tumour and D) extracranial solid tumour. We compared the monthly observed ASIRs in the pandemic period of March to November 2020 with the projected hypothetical continuation of the trends in the prepandemic period of March 2016 to February 2020.
Observed and projected monthly age-standardized incidence rates (ASIRs) for all pediatric cancers combined per million, by geographic area: A) Atlantic provinces (Nova Scotia, Prince Edward Island, New Brunswick and Newfoundland and Labrador), B) Quebec, C) Ontario, D) Prairie provinces (Manitoba, Saskatchewan and Alberta) and E) British Columbia. We compared the monthly observed ASIRs in the pandemic period of March to November 2020 with the projected hypothetical continuation of the trends in the prepandemic period of March 2016 to February 2020.
Table 3 shows that the percentage of patients enrolled in clinical trials was 10.7% between March and May 2020, 14.0% between June and August 2020, and 15.0% between September and November 2020. The median enrolment rate was 8.8% (IQR 7.0% to 9.5%) during the baseline period. There was a significant increase in enrolment in clinical trials during the third quarter of the pandemic (RR 1.71, 95% CI 1.01 to 2.89) compared to baseline, which was largely attributable to an increase in enrolment rate for leukemia and lymphoma during that period (Table 2).
Proportion of children enrolled in a clinical trial, with metastatic disease at cancer diagnosis and who died within 30 days
For each pandemic quarter, 19.1%, 27.7% and 22.5%, respectively, of patients presented with metastatic disease, which was comparable to baseline overall (median 22.7%, IQR 20.5% to 24.9%) and stratified by cancer type (Table 3). Similarly, the proportion of patients with leukemia who presented with CNS involvement or NCI high-risk disease was comparable to baseline (Table 3).
The percentage of patients who died within 30 days of presentation was 2.1% between March and May 2020, 0.8% between June and August 2020 and 0.0% between September and November 2020, which was comparable to the baseline rate (median 1.2%, IQR 0.8% to 1.3%). Analyses using a weighted model (time-dependent variance) rather than an unweighted model (using constant variance) yielded similar results (data not shown).
Interpretation
We did not find a statistically significant difference in the incidence of new cancer diagnoses in Canadian children aged younger than 15 years during the first 9 months of the COVID-19 pandemic compared with previous years. In addition, we observed no significant differences in the proportion of patients enrolled in a clinical trial, presenting with metastatic disease or who died within 30 days of presentation.
Our findings suggest that among children in Canada, cancer diagnosis was not delayed during the pandemic, unlike findings described in previous reports.10–13 Although access to emergency departments markedly dropped during the pandemic, there may have been less reluctance by families and health care professionals to access health care for serious symptomatology.33,34 Furthermore, most newly diagnosed patients in Canada have access to publicly funded health care, lessening the impact of financial hardship. Alternatively, it may also suggest that delays were sufficiently short that they did not affect cancer incidence rates when calculated monthly. Of note, we did not observe a reduction in new cases of extracranial solid tumours or CNS tumours; diagnosis of these cancers is dependent on adequate access to diagnostic imaging and surgical procedures. We studied the first 9 months of the pandemic; it is possible that changes in cancer detection and outcomes will be appreciable only in the longer term.
Our findings regarding stable enrolment in clinical trials also diverge from the experiences of other researchers.35–39 For example, the Innovative Therapies for Children with Cancer consortium in Europe reported a profound disruption in pediatric early clinical research owing to a reduction in personnel, cancellation of monitoring visits and difficulty with sample transportation,36 which led to a 75% reduction in enrolment.37 Our results mostly reflect enrolment in phase III clinical trials led by academic consortia and are probably not generalizable to enrolment in early phase or translational trials that may require travel to quaternary hospitals or may not have been prioritized by institutions because of resource constraints. The ongoing recruitment in clinical trials that we observed might be explained by enrolment in therapeutic trials being considered an integral component of high-quality care in pediatric cancer22 and thus being prioritized by institutions. It is also possible that the impact of the COVID-19 pandemic was minimized by the proactive and extensive changes established by some research consortia and funding agencies, such as flexibility for patients’ enrolment and follow-up schedules, and implementation of remote monitoring visits,35 which could benefit children and their families as the pandemic wanes.
Limitations
Interpretation of our findings requires consideration of some limitations. The CYP-C collects data only from pediatric oncology centres. It is possible that some children are not yet properly referred to tertiary centres. However, this would be more concerning if new diagnoses had decreased. External factors unrelated to the COVID-19 pandemic might affect enrolment in clinical trials, given that the availability of clinical trials varies over time. For example, accrual in the Children’s Oncology Group front-line trials for acute B-cell lymphoblastic leukemia started in 2020 for several Canadian hospitals and may partly explain the increase in clinical trial enrolment for patients with leukemia. Moreover, estimates of enrolment rate may be unstable given the relatively small number of patients and clinical trials. Some positive findings could be attributed to type I errors caused by the large number of comparisons without adjustment for multiplicity. Finally, the study follow-up period does not allow the full range of the possible negative impacts of the COVID-19 pandemic to be understood. Our data likely did not capture adjustment of treatments such as delays in chemotherapy, radiotherapy or hematopoietic stem cell transplantation. As subsequent waves of COVID-19 cases unfold in Canada and abroad, continued surveillance is necessary to understand the ramifications of the extensive changes made in response to the COVID-19 pandemic, such as reorganization of care and more generalized use of telehealth.
Conclusion
We did not observe a statistically significant change in the ASIRs of childhood cancer during the first 9 months of the COVID-19 pandemic in Canada compared with the period before the pandemic. Moreover, enrolment in clinical trials remained stable, and we did not observe an increase in the proportion of patients with metastatic disease or early mortality. Although these results are reassuring, continued surveillance is necessary to ascertain potential long-term negative effects of the COVID-19 pandemic among children with cancer.
Acknowledgements
The authors are grateful for the contributions of study participants, participating pediatric oncology centres, members of the Cancer in Young People in Canada (CYP-C) Management and Advisory Committees and the Pediatric Oncology Group of Ontario. The authors also thank all data managers and principal investigators at the 17 CYP-C sites for their collaboration and dedicated work.
Footnotes
Competing interests: None declared.
This article has been peer reviewed.
Contributors: All authors contributed to research design and data interpretation. Lin Xie, Marie-Claude Pelland-Marcotte, Sulaf Elkhalifa, Mylene Frechette, Jaskiran Kaur and Jay Onysko were involved in data analysis. Marie-Claude Pelland-Marcotte prepared the first version of this manuscript. All of the authors reviewed the manuscript critically for important intellectual content, gave final approval of the version to be published and agreed to be accountable for all aspects of the work.
Funding: Cancer in Young People in Canada is funded by the Public Health Agency of Canada. No additional funding was provided for this study. Lillian Sung is supported by the Canada Research Chair in Pediatric Oncology Supportive Care.
Data sharing: As per Cancer in Young People in Canada policies, individual participant data will not be shared. Application for utilization of data can be submitted through the C17 Council website (available at http://www.c17.ca/index.php?cID=70 [login required]).
Disclaimer: Data used in this publication are from the Cancer in Young People in Canada Surveillance Program and are used with the permission of the Public Health Agency of Canada.
- Accepted October 4, 2021.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use), and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/